NADH
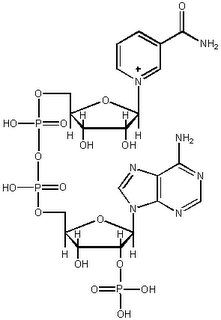
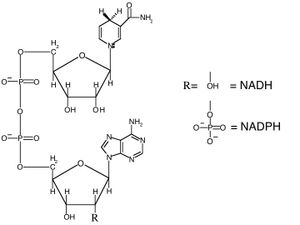 Nicotinamide adenine dinucleotide
Nicotinamide adenine dinucleotideis an important cofactor. NADH is the reduced form, while NAD+ is the oxidized form. Phosphorylation through an ester linkage at the 2' position of the adenosyl nucleotide yields NADP.
Phototrophs obtain NADPH (along with ATP) through the noncyclic fom of photophosphorylation during the light-reactions of photosynthesis (image Z-scheme). A photon is absorbed by Photosystem II and the two resultant excited electrons are passed to Photosystem I, which employs a second photon to further boost their energy for the overall reaction: NADP+ + H+ + 2e- → NADPH. Non-phototrophic organisms manufacture NADH during oxidative phosphorylation, where NADH & FADH2, the energy carrier molecules generated by the citric acid cycle enter an electron transfer chain that generates a proton gradient by pumping protons (H+) across the membrane. Glycolysis generates the energy carrier molecules ATP and NADH in addition to intermediates (3-C or 6-C) for biosynthetic pathways.
Nicotinamide adenine dinucleotide phosphate (NADP) is employed in anabolic reactions, such as fatty acid biosynthesis and nucleic acid synthesis, which require NADPH as a reducing agent.
In chloroplasts, the light-reactions of noncyclic photophosphorylation produce both ATP and NADPH, which act as energy-transfer, reducing agents in the light-independent (“dark”) reactions of the Calvin cycle.
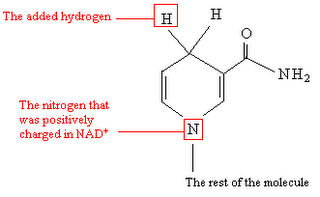
MH2 + NAD+ → NADH + H+ + M: + energy, where M is a metabolite.
Two hydrogen ions (a hydride ion and an H+ ion) are transferred from the metabolite. One electron is transferred to the positively-charged nitrogen, and one hydrogen attaches to the carbon atom opposite to the nitrogen.
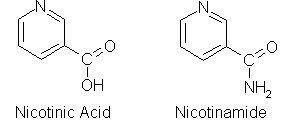
NAD is synthesized from the vitamin niacin in the form of nicotinic acid or nicotinamide. At right is adenine.
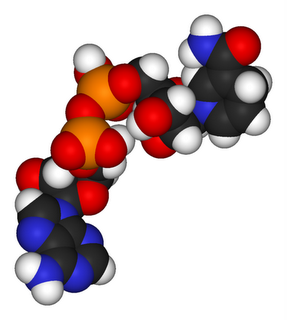
Left: space fill molecule of NADH
C black : H white : O red : P orange : N blue








































0 Comments:
Post a Comment
<< Home